The atropine landscape is constantly evolving as new studies are published, and it can be difficult as a clinician to make decisions about what concentrations provide the highest myopia control efficacy but lowest side effect profile for your patient. Not to mention keeping abreast of all the catchy acronyms! Here is a compiled summary of all of the most recent atropine studies, which you can then apply to your clinical decision making. There’s also some information on trials underway, and finally the STAAR study which is recruiting right now. Click on any of the headings to link to the relevant publication.
THE LATEST PUBLISHED STUDIES

LAMP: Low-concentration atropine for myopia progression (LAMP): A randomized, double-blinded, placebo-controlled trial of 0.05%, 0.025% and 0.01% atropine eye drops in myopia control
Jason Yam,Yuning Jiang等人在2019年发表了《灯研究》,该研究在香港针对438名儿童进行。该研究的目的是发现0.01%是否是近视控制的有效浓度,以及最佳的进展浓度。它有四个建议的阶段;
- 阶段1:安慰剂的治疗组1年,0.05%,0.025%和0.01%
- 阶段2:安慰剂组更改为第1阶段确定的最高疗效组,其他组持续了一年
- Phase 3: One year washout period for all except the change-over group
- Phase 4: Atropine resumed in children whom have progressed more than 0.5D in Phase 3, and change-over group continued without pause.
本文(从标题链接)提供了第1阶段的结果,其中至少为-1.00d,在过去一年中至少取得了至少-0.5d的效果,其中有438名受试者。与安慰剂相比,所有三个浓度均显示出近视进展的降低,并取决于浓度。在一年中,平均0.05%的组显示-0.27D变化(减少66%),0.025%组显示-0.46D变化(降低43%),0.01%组显示-0.59D变化(-0.59D变化(降低27%),最后安慰剂组的平均变化为-0.81d。然而,值得注意的是,轴向长度(Al)组之间没有统计学差异,这表明这是无效的。相比之下,与对照组相比,0.05%的轴向伸长率降低了51%和0.025%,增长了29%。轴向和折射结局与阿托品的不匹配意味着我们必须看到轴向长度在任何研究中都会导致并发对照组正确理解疗效。
All of the concentrations were tolerated, very few children required progressive addition spectacles (the same across all groups) and there were no medication reported adverse events. This study is only in its first phase though, and as shown by ATOM2 the results can change in the second year. Stay tuned.
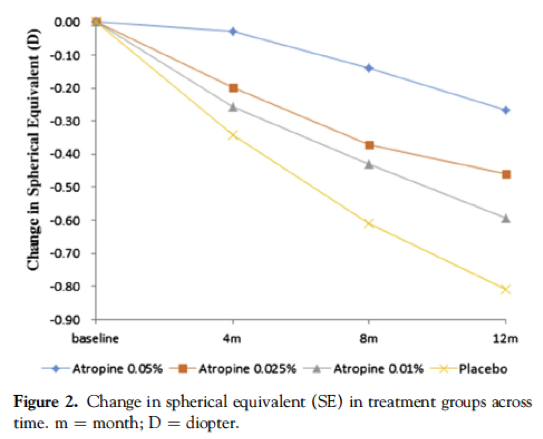
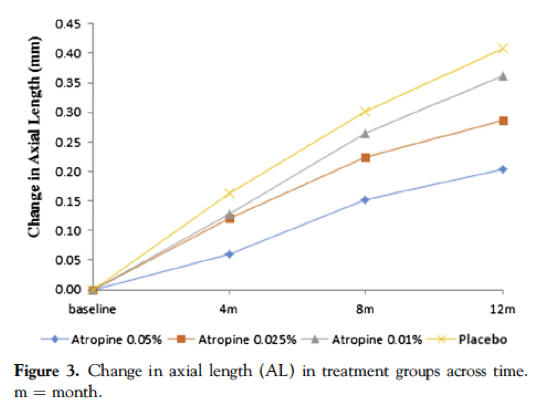
These figures have been reproduced from the paper, which is open access.
Take home message:0.05% was the most effective, and 0.01% showed no statistically significant slowing in axial length compared to placebo. All concentrations were tolerated including the 0.05%, over a one year period.

灯阶段2结果 - 第二年
Hot off the Press: in fact 'In Press' at the time of writing! In Phase 2, the first year's placebo group was changed to 0.05% atropine treatment for a year, while the children undergoing treatment with 0.01%, 0.025% and 0.05% in the first year continued with the same treatment across a second year. 383 of the original 438 children completed two years of follow up.
Over the 2-year period, the mean SE progression was a mean of 0.55D / 0.39mm, 0.85D / 0.50mm, and 1.12D / 0.59mm in the 0.05%, 0.025%, and 0.01% atropine groups, respectively. Without a control group in this second year, percentage reductions cannot be calculated.
Compared with the first year, the second-year efficacy of 0.05% and 0.025% atropine remained similar, but improved mildly in the 0.01% atropine group. For the group changed from placebo in year one to 0.05% atropine in year two, their myopia progression was reduced from 0.82D / 0.43mm in the first year to 0.18D / 0.15mm in the second year. The effects on accommodation (loss of 0.3 to 1.6D amps) and pupil size (increase by up to 1mm) in all concentrations was similar in the second year to the first year - for more on this see our summary blogAtropine -wonder or weak treatment?Visual acuity and vision-related quality of life remained unaffected.
Take home message:Over 2 years, the efficacy of 0.05% atropine observed was around double that observed with 0.01% atropine. Starting previously untreated children on 0.05% atropine showed a significant result and it was well tolerated in this population of Hong Kong Chinese children.

低剂量阿托品对近视进展,瞳孔直径和适应性振幅的影响:低剂量阿托品和近视进展
发表在2019年10月由Aicun有趣,菲奥娜堵塞leton and others, 400 children were randomly assorted to groups using 0.01% atropine or 0.02% atropine for twelve months. Children were between 6-14 years, had myopia of more than -1.00, but prior progression was not an inclusion criteria. An atropine concentration-dependent response was shown amongst the children after one year, statistically similar in both myopic change and axial length. Refractive change was on average -0.38D in 0.02% (45% reduction), -0.47D in 0.01% (33% reduction) and -0.70D in the placebo. Axial length increased 0.46mm in the control group, 0.37mm (20% reduction) in the 0.01% group and 0.30mm(35% reduction) in the 0.02% group.
The authors found that both atropine concentrations reduced accommodative amplitude by around 1.5D and increased pupil size by less than 1mm compared to the control group, but there was no difference between the two concentrations.
Take home message:0.02%阿托品的耐受性良好,具有相似的副作用与0.01%,但表现出更好的功效。

The diluted atropine for inhibition of myopia progression in Korean Children
Ji-Sun Moon and Sun Young Shin examined 285 children aged 5-14 years with less than 6D of myopia, and assigned them to 0.01%, 0.025% and 0.05% atropine dosage groups based on how fast they were progressing, for one year.
According to their protocol, the 0.05% atropine was prescribed when the calculated myopia progression rate exceeded -1.50 D/y, the 0.025% atropine was prescribed when progression was between -1.00 D/y and less than -1.50 D/y, and 0.01% atropine was prescribed when the calculated myopia progression was below -1.00 D/y. This means it wasn't a randomized study, and there was no control group.
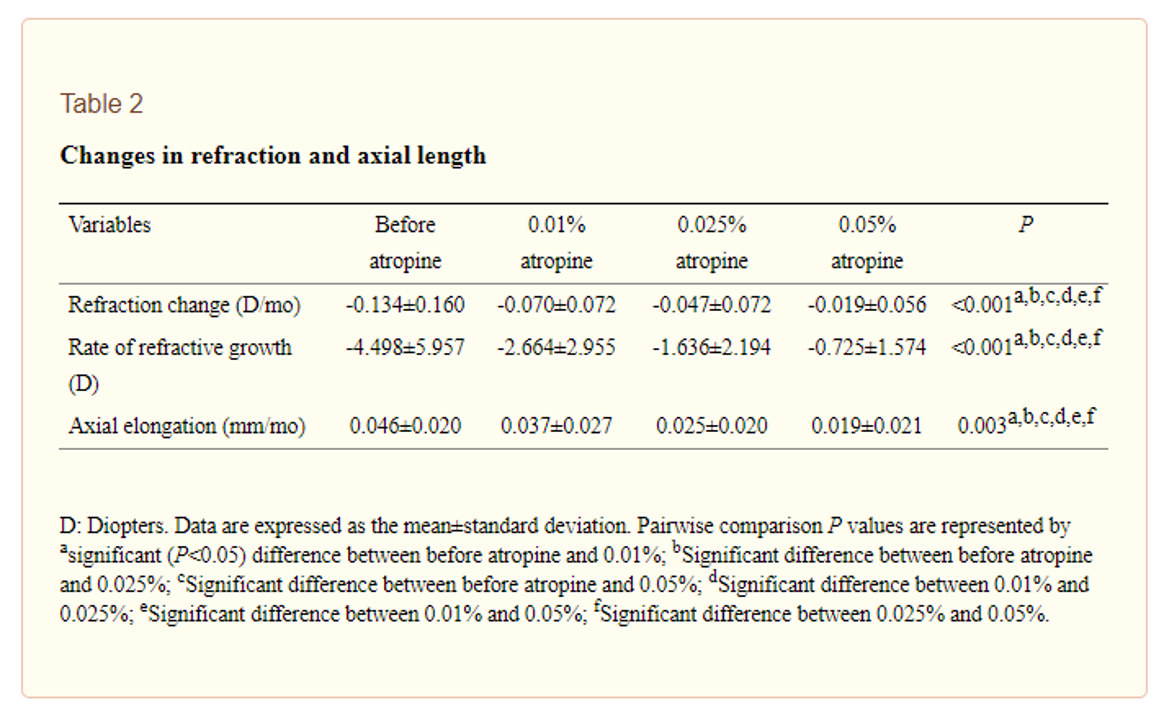
他们使用的阿托品从市售的1%阿托品中稀释。有趣的是,浓度对瞳孔大小和适应性振幅的影响没有差异(俯卧撑测试) - 实际上,与“阿托普汀之前”处理的测量相比,这两种测量都没有显着变化。如果没有像其他研究那样典型的副作用,这使一个思考稀释的处理的稳定性如何。但是,仍然存在浓度依赖性的响应 - 请参见下面的表2从开放式访问纸中复制 - “阿托品治疗前”和增加的浓度之间发现了显着差异。与0.01%的组相比,更快的进步者在0.05%的组中,这一点尤其令人着迷,但近视控制结果最好。
Over the one year study, the myopia progression in the 0.01% was -0.84D, in 0.025% -0.56D and in 0.05% -0.23D. Prior to study entry, children were on average progressing 1.61D per year. There were more adverse events in the higher concentration groups, but not enough to be statistically significant.
Take home message:Hmmm. There's no control group, it wasn't randomized, and comparing a prior progression rate to the rate after treatment, in the same person, is spurious as progression tends to slow with time anyway. It does appear again, though, that the concentration-dependent efficacy of atropine has been demonstrated.

Efficacy and Safety of 1% Atropine on Retardation of Moderate Myopia Progression in Chinese School Children
Zhu et al introduced in January 2020 a paper on controlling Myopia with 1% atropine. Concerned with the “rebound” effects demonstrated in the ATOM-5 year follow up, this time the atropine was delivered differently, and reduced in three stages. 660 children were initially enrolled in the study, with myopia of -2.00D to -8.00D.
Initially, phase I, the children received one drop at bedtime, once a month for twenty-four months. In the second year, phase II, drops were reduced to once every two months. Phase III included no drops and the children were monitored for a year. Side effects were treated by progressive lenses with transition. The Atropine did slow the progression, as expected, with the treatment group progressing on average -0.50D over the two years, when the control group progressed almost -2.00D over the study. There was also no “rebound” effect produced during phase III, and the treatment group progressed not only less overall, but during the phase III where no atropine treatment was occurring.
What this means for your clinical practice:This is also a novel dosing regime, with drops only being instilled once a month, which changes our conventional thinking on atropine. The atropine 1% side effects are high, with a large rate of children in the study affected, which will impact part of your treatment decisions if proceeding with 1%, but the convenience of only having to dose once a month is also relevant. This may be an appropriate approach for some of your patients.
STUDIES CURRENTLY UNDERWAY

Low-dose (0.01%) atropine eye-drops to reduce progression of myopia in children: a multicentre placebo-controlled randomised trial in the UK (CHAMP-UK).
The CHAMP study is currently underway in the UK, involving 289 children aged 6-12 years. In a double-masked study design, children will be randomly allocated to 0.01% or placebo, at a 2:1 ratio, and reviewed every six month. The duration is two years, with the primary outcome of cycloplegic autorefraction, however they also plan on measuring axial length and other parameters. After two years, atropine treatment will be ceased, and a follow up conducted three years later to evaluate refractive error and adverse events. The estimated primary completion date is December 2021.
The Low-Dose Atropine for Treatment of Myopia: By the Paediatric eye disease investigator group (PEDIG).
[Link to clinical trial protocol]
American children aged 5-13 years will be allocated to either atropine 0.01% or a placebo for two years in this multi-site study. The outcomes for six months post cessation will also be measured. They’ve enrolled 187 participants with the primary measurements being the degree of spherical equivalent refractive error as well as axial length. Inclusion criteria is myopia -1.00 to -6.00, but no restrictions on previous progression. Estimated completion date is September 2022.

The Myopia outcome study of atropine in children (MOSAIC)
250 children aged 6-16 years were recruited in Ireland if they had “progressive myopia.” They were randomised to two groups, 0.01% atropine or placebo. This study will be investigating a proprietary non-preserved atropine formulation developed by内瓦卡,主要是高加索人口。
经过两年的监测后,治疗组将进行12个月的冲洗期,安慰剂组将在跨界设计中分配到0.01%的阿托品中12个月。他们还打算在过去36个月中跟进,具体取决于最初的结果。主要测量是球形折射率,但是它们还打算测量轴向长度和从0.01%的任何潜在反弹。该研究预计将于2023年5月完成。
在儿童近视研究nvk - 002(冠军)
[Link to clinical trial protocol]
内瓦卡is additionally trialling their new commercially prepared, preservative free low-dose atropine formulation in an American multicentered, randomized, double-masked, vehicle-controlled study. Children 3-17 years old with myopia of -0.50 to -6.00 have been randomly allocated to one of three groups, NVK-002 low concentration, NVK-002 high concentration and placebo. Stage 1 of the trial extends for three years, where the groups are measured for spherical equivalent refractive error. Stage 2 involves random re-allocation of the groups to a second group (except for placebo, who will be allocated to a treatment group). As this is a new formulation/manufacturer the safety and any adverse effects is also being monitored. Study completion date is estimated to be August 2023.
The safety and efficacy of SYD-101 in children with Myopia (STAAR)
[Link to clinical trial protocol]
This study is looking at a new formulation of pre-prepared atropine made bySydnexis, using proprietary technology designed for maximum stability and accurate dosing. SYD-101 is hoping to have a longer shelf life, and higher tolerability than previously tested commercially prepared atropine formulas. 840 children with myopia of -0.50 to -6.00 that are 3 to 14 are being recruited in America. They will be randomly assigned to one of three groups, SYD-101 Dose 1, SYD-101 Dose 2 and placebo. Then after three years, participants will be re-assigned to another group for one year, ensuring the original placebo group is assigned to a SYD-101 group. The primary outcome is looking for myopic progression greater than 0.75D, but also looking at the mean annual myopic progression, time to progression of 0.75D and the mean change in axial length. This study is recruiting currently, with 46 locations across the United States - clickthis ClinicalTrials.gov linkto see a list of sites and a contact email if you want to learn more.
Microdosed Atropine 0.1% and 0.01% Ophthalmic Solutions for Reduction of Pediatric Myopia Progression
[Link to clinical trial protocol]
2019年底,已经开始了一项新的试验,研究了由微剂量原子剂提供的不同浓度的阿托品的影响。该研究由SUNY验光学院进行,并由Eyenovia资助,将比较0.1%,0.01%和安慰剂剂量。阿托品将由微剂量分配器传递,它们的理论将降低全身副作用和总体耐受性。将为儿童提供3年的初始解决方案,然后再将其重新分配给另一组,持续十二个月。
This study may show that a novel dispensing mechanism may make 0.1% atropine more tolerable, and add to our body of evidence as to which is the ultimate concentration of atropine to adequately control progressing myopia. This study is currently accepting new participants, between the ages of 3-12 whom are between -1.00 and -6.00 at various sites across the USA.
Read more of our six-part series on atropine
何时为近视控制的阿托品开处方
阿托品复合的并发症
Complex atropine cases
Systemic side effects of atropine eye drops
阿托品 - 奇迹还是弱治疗?
Check out these clinical cases
Which atropine dosage should I prescribe for myopia control?
Atropine eye drops and ocular allergy - what's the cause?
You can also listen to our three podcasts on atropine with world-leading researchers
阿托品,与Karla Zadnik教授参与科学和负责任的实践from Ohio State University, USA.
More on atropine 0.01% treatment for myopia management with Professor Mark Bullimorefrom the University of Houston, Texas USA.
Atropine 0.01% for myopia management with Professor James Loughmanfrom Technological University Dublin, and the Centre for Eye Research Ireland.

About Cassandra
Cassandra Haines是一个临床验光师、研究员和作家with a background in policy and advocacy from Adelaide, Australia. She has a keen interest in children's vision and myopia control.
由于不受限制的教育赠款,这些教育内容是给您带给您的








