阿托品景观也在不断地演变为new studies are published, and it can be difficult as a clinician to make decisions about what concentrations provide the highest myopia control efficacy but lowest side effect profile for your patient. Not to mention keeping abreast of all the catchy acronyms! Here is a compiled summary of all of the most recent atropine studies, which you can then apply to your clinical decision making. There’s also some information on trials underway, and finally the STAAR study which is recruiting right now. Click on any of the headings to link to the relevant publication.
THE LATEST PUBLISHED STUDIES

灯:近视进展的低浓度阿托品(LAMP):一项随机,双盲,安慰剂对照试验在肌病控制中为0.05%,0.025%和0.01%阿托品眼滴
Jason Yam, Yuning Jiang and others in 2019 published the LAMP study, conducted in Hong Kong on 438 children. The study was aiming to discover if 0.01% is an effective concentration for myopia control, along with the optimal concentration for slowing progression. It has four proposed stages;
- Phase 1: 1 year of treatment groups of placebo, 0.05%, 0.025% and 0.01%
- Phase 2: Placebo group is changed to the highest efficacy group determined in Phase 1, and the other groups continue for one year
- Phase 3: One year washout period for all except the change-over group
- Phase 4: Atropine resumed in children whom have progressed more than 0.5D in Phase 3, and change-over group continued without pause.
This article (linked from the heading) provides the results of Phase 1, of children whom are at least -1.00D and had progressed at least -0.5D in the past year, with 438 subjects enrolled. All three concentrations were shown to cause reduced myopia progression compared to the placebo, with a concentration dependent result. At one year, on average, the 0.05% group showed a -0.27D change (66% reduction), the 0.025% group showed a -0.46D change (43% reduction), the 0.01% group showed a -0.59D change (27% reduction) and finally the placebo group had an average change of -0.81D. Of note however, there was not statistical difference in the axial length (AL) retardation between 0.01% group and the placebo, suggesting it was not effective. By comparison, 0.05% slowed axial elongation by 51% and 0.025% by 29% compared to the control group. This mismatch between axial and refractive outcomes with atropine means that we must see axial length results in a concurrent control group, in any study, to properly understand efficacy.
All of the concentrations were tolerated, very few children required progressive addition spectacles (the same across all groups) and there were no medication reported adverse events. This study is only in its first phase though, and as shown by ATOM2 the results can change in the second year. Stay tuned.
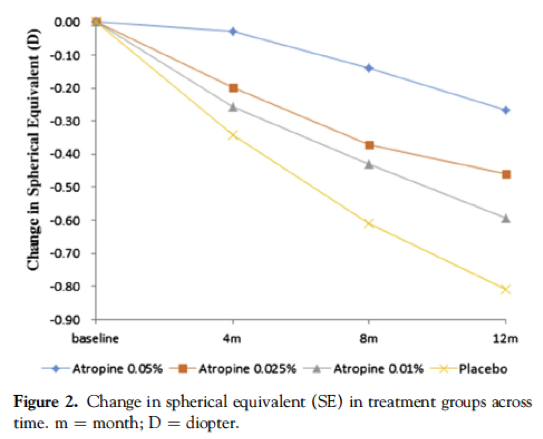
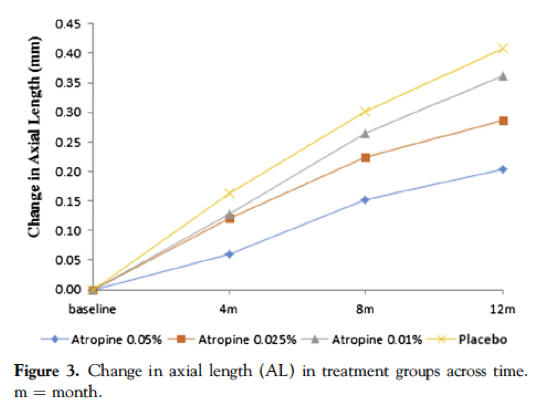
These figures have been reproduced from the paper, which is open access.
Take home message:0.05% was the most effective, and 0.01% showed no statistically significant slowing in axial length compared to placebo. All concentrations were tolerated including the 0.05%, over a one year period.

LAMP Phase 2 results - the second year
Hot off the Press: in fact 'In Press' at the time of writing! In Phase 2, the first year's placebo group was changed to 0.05% atropine treatment for a year, while the children undergoing treatment with 0.01%, 0.025% and 0.05% in the first year continued with the same treatment across a second year. 383 of the original 438 children completed two years of follow up.
在两年期间,平均SE进展分别为0.55D / 0.39mm,0.85D / 0.50mm和1.12d / 0.59mm的平均值,分别为0.05%,0.025%和0.01%的Atropine组。在第二年,如果没有对照组,就无法计算降低百分比。
Compared with the first year, the second-year efficacy of 0.05% and 0.025% atropine remained similar, but improved mildly in the 0.01% atropine group. For the group changed from placebo in year one to 0.05% atropine in year two, their myopia progression was reduced from 0.82D / 0.43mm in the first year to 0.18D / 0.15mm in the second year. The effects on accommodation (loss of 0.3 to 1.6D amps) and pupil size (increase by up to 1mm) in all concentrations was similar in the second year to the first year - for more on this see our summary blog阿托品-wonder or weak treatment?Visual acuity and vision-related quality of life remained unaffected.
Take home message:Over 2 years, the efficacy of 0.05% atropine observed was around double that observed with 0.01% atropine. Starting previously untreated children on 0.05% atropine showed a significant result and it was well tolerated in this population of Hong Kong Chinese children.

Effect of low-dose atropine on myopia progression, pupil diameter and accommodative amplitude: Low dose atropine and myopia progression
Published in October 2019 by Aicun Fun, Fiona Stapleton and others, 400 children were randomly assorted to groups using 0.01% atropine or 0.02% atropine for twelve months. Children were between 6-14 years, had myopia of more than -1.00, but prior progression was not an inclusion criteria. An atropine concentration-dependent response was shown amongst the children after one year, statistically similar in both myopic change and axial length. Refractive change was on average -0.38D in 0.02% (45% reduction), -0.47D in 0.01% (33% reduction) and -0.70D in the placebo. Axial length increased 0.46mm in the control group, 0.37mm (20% reduction) in the 0.01% group and 0.30mm(35% reduction) in the 0.02% group.
作者发现,与对照组相比,两种阿托品浓度都将可容纳幅度降低了1.5D左右,而学生的大小则小于1mm,但是两种浓度之间没有差异。
Take home message:0.02% atropine is well tolerated, had a similar side effect profile to 0.01% but showed better efficacy.

The diluted atropine for inhibition of myopia progression in Korean Children
Ji-Sun Moon and Sun Young Shin examined 285 children aged 5-14 years with less than 6D of myopia, and assigned them to 0.01%, 0.025% and 0.05% atropine dosage groups based on how fast they were progressing, for one year.
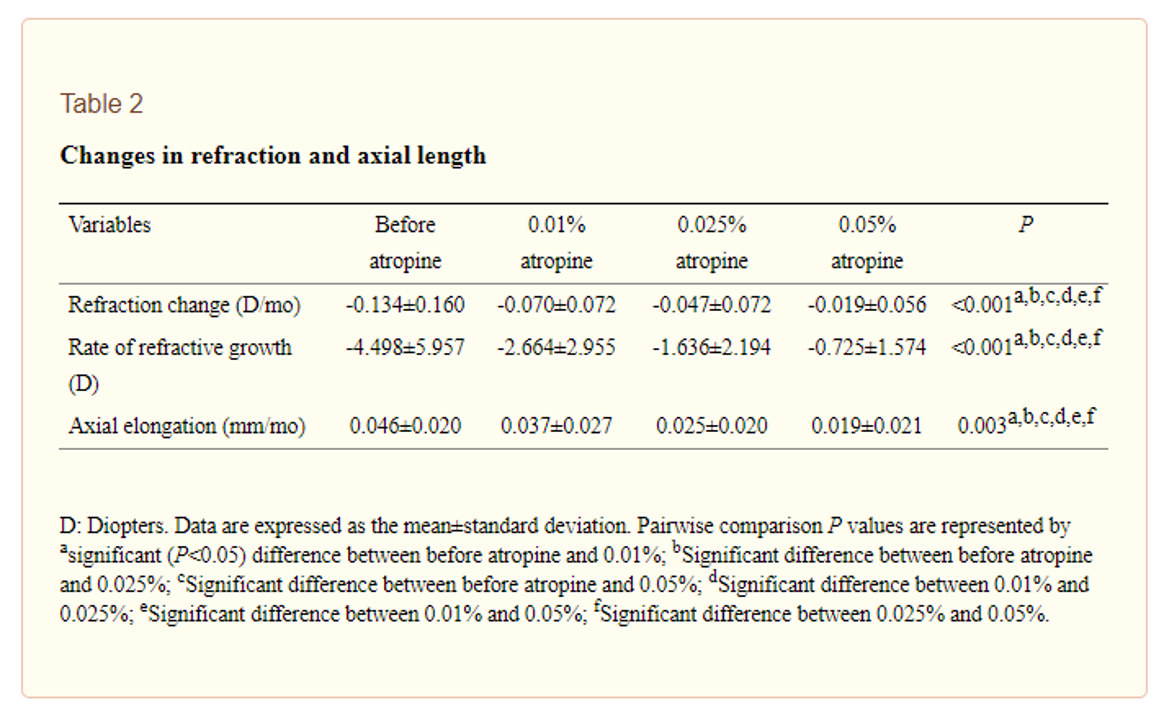
根据他们的方案,当计算得出的近视进度率超过-1.50 d/y时,规定了0.05%的阿托品,当进展为-1.00 d/y且小于-1.50 d/y时,规定了0.025%阿托品当计算出的近视进展低于-1.00 d/y时,处方了阿托品%。这意味着这不是一项随机研究,也没有对照组。
他们使用阿托品稀释从商业ly available 1% atropine. Interestingly, there was no difference in the effect of the concentrations on pupil size and accommodative amplitude (push up test) - in fact, no significant change in either from measurements taken 'before atropine' treatment. This makes one ponder how stable the diluted treatments were, if no side effects were measured as is typical in other studies. However there was still a concentration-dependent response - see Table 2 below reproduced from the open access paper - significant differences were found between 'before atropine' treatment and the increasing concentrations seemed to work better, in turn. This is particularly fascinating as the faster progressors were in the 0.05% group by comparison to the 0.01% group, yet had the best myopia control result.
Over the one year study, the myopia progression in the 0.01% was -0.84D, in 0.025% -0.56D and in 0.05% -0.23D. Prior to study entry, children were on average progressing 1.61D per year. There were more adverse events in the higher concentration groups, but not enough to be statistically significant.
Take home message:Hmmm. There's no control group, it wasn't randomized, and comparing a prior progression rate to the rate after treatment, in the same person, is spurious as progression tends to slow with time anyway. It does appear again, though, that the concentration-dependent efficacy of atropine has been demonstrated.

Efficacy and Safety of 1% Atropine on Retardation of Moderate Myopia Progression in Chinese School Children
Zhu et al introduced in January 2020 a paper on controlling Myopia with 1% atropine. Concerned with the “rebound” effects demonstrated in the ATOM-5 year follow up, this time the atropine was delivered differently, and reduced in three stages. 660 children were initially enrolled in the study, with myopia of -2.00D to -8.00D.
最初,第一阶段,孩子们每月一次下降一次,持续二十四个月。在第二年,第二阶段的下降减少到每两个月一次。第三阶段不包括滴水,对孩子进行了一年的监控。通过过渡的进行性镜头处理副作用。正如预期的那样,阿托品确实减慢了进展,而治疗组在两年中平均进展为-0.50d,当时对照组在研究中取得了近-2.00d的进展。在第三阶段期间也没有产生“反弹”效应,并且治疗组的整体进展不少,而且在没有发生阿托品治疗的第三阶段。
What this means for your clinical practice:这也是一种新颖的剂量制度,每月只能灌输一次,这改变了我们对阿托品的传统思维。阿托品1%的副作用很高,研究中的儿童率很大,如果以1%的速度继续进行治疗的决定,则会影响您的一部分治疗决策,但是每月只需服用一次的便利性也很重要。对于您的某些患者来说,这可能是一种合适的方法。
STUDIES CURRENTLY UNDERWAY

Low-dose (0.01%) atropine eye-drops to reduce progression of myopia in children: a multicentre placebo-controlled randomised trial in the UK (CHAMP-UK).
The CHAMP study is currently underway in the UK, involving 289 children aged 6-12 years. In a double-masked study design, children will be randomly allocated to 0.01% or placebo, at a 2:1 ratio, and reviewed every six month. The duration is two years, with the primary outcome of cycloplegic autorefraction, however they also plan on measuring axial length and other parameters. After two years, atropine treatment will be ceased, and a follow up conducted three years later to evaluate refractive error and adverse events. The estimated primary completion date is December 2021.
The Low-Dose Atropine for Treatment of Myopia: By the Paediatric eye disease investigator group (PEDIG).
American children aged 5-13 years will be allocated to either atropine 0.01% or a placebo for two years in this multi-site study. The outcomes for six months post cessation will also be measured. They’ve enrolled 187 participants with the primary measurements being the degree of spherical equivalent refractive error as well as axial length. Inclusion criteria is myopia -1.00 to -6.00, but no restrictions on previous progression. Estimated completion date is September 2022.

儿童阿托品的近视结果研究(马赛克)
250 children aged 6-16 years were recruited in Ireland if they had “progressive myopia.” They were randomised to two groups, 0.01% atropine or placebo. This study will be investigating a proprietary non-preserved atropine formulation developed byNevakar, in a primarily Caucasian population.
After two years of monitoring, the treatment group will undergo a washout period for 12 months and the placebo group will be assigned to 0.01% atropine for 12 months, in a crossover design. They also intend to follow up past 36 months, depending on initial outcomes. Primary measurement is the spherical refractive error, however they also intend to measure axial length and any potential rebound from the 0.01%. The study is expected to be completed in May 2023.
Study of NVK-002 in Children with myopia (CHAMP)
Nevakar另外,在美国多中心,随机,双掩盖的车辆对照研究中,试验了他们新的商业准备的,防腐剂的无剂量阿托品制剂。近视为-0.50至-6.00的3-17岁儿童已随机分配给三组之一,NVK-002低浓度,NVK-002高浓度和安慰剂。该试验的第1阶段延长了三年,在该阶段测量了球形等效折射率。第2阶段涉及将小组随机重新分配给第二组(安慰剂除外,将分配给治疗组)。由于这是一种新的配方/制造商,因此安全性和任何不利影响也将受到监控。研究完成日期估计为2023年8月。
安全性和efficacy of SYD-101 in children with Myopia (STAAR)
这项研究正在研究由预先准备的阿托品制定的新配方Sydnexis, using proprietary technology designed for maximum stability and accurate dosing. SYD-101 is hoping to have a longer shelf life, and higher tolerability than previously tested commercially prepared atropine formulas. 840 children with myopia of -0.50 to -6.00 that are 3 to 14 are being recruited in America. They will be randomly assigned to one of three groups, SYD-101 Dose 1, SYD-101 Dose 2 and placebo. Then after three years, participants will be re-assigned to another group for one year, ensuring the original placebo group is assigned to a SYD-101 group. The primary outcome is looking for myopic progression greater than 0.75D, but also looking at the mean annual myopic progression, time to progression of 0.75D and the mean change in axial length. This study is recruiting currently, with 46 locations across the United States - click这个临床。gov链接如果您想了解更多信息,请查看网站列表和联系电子邮件。
Microdosed Atropine 0.1% and 0.01% Ophthalmic Solutions for Reduction of Pediatric Myopia Progression
A new trial has been launched in late 2019 looking at the effects of different concentrations of atropine, delivered by a microdosing atomiser. Conducted by the SUNY School of Optometry, and funded by Eyenovia, the study will compare 0.1%, 0.01% and a placebo dose. The atropine will be delivered by a microdose dispenser, which they theorise will reduce systemic side effects and overall tolerance. Children will be given the initial solution for 3 years, then re-allocated to another group for a further twelve months time.
这项研究可能表明,一种新型的分配机制可能会使0.1%阿托品更耐受,并增加我们的证据,表明阿托品的最终浓度可以充分控制近视的近视。这项研究目前正在接受新的参与者,年龄在3-12岁之间,他们在美国各个地点之间在-1.00至-6.00之间。
Read the rest of our six-part series on atropine
Check out these clinical cases
You can also listen to our three podcasts on atropine with world-leading researchers
阿托品, engaging with science and responsible practice with Professor Karla Zadnikfrom Ohio State University, USA.
More on atropine 0.01% treatment for myopia management with Professor Mark Bullimorefrom the University of Houston, Texas USA.
阿托品0.01% for myopia management with Professor James Loughmanfrom Technological University Dublin, and the Centre for Eye Research Ireland.
This educational content is brought to you thanks to unrestricted educational grant from
















